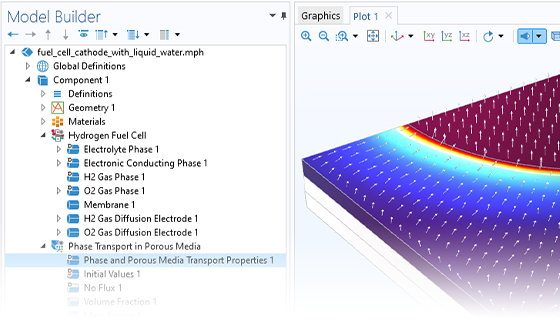
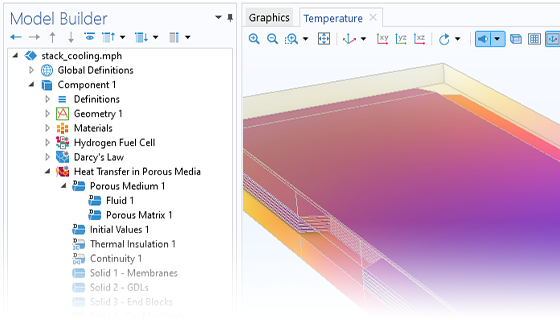
Hydrogen Fuel Cells
The Fuel Cell & Electrolyzer Module features predefined formulations for the most common types of hydrogen fuel cells and accounts for the electrodes, electrolyte, as well as current collectors and feeders. Examples of fuel cell types that can be modeled are PEMFC, AFC, PAFC, SOFC, MCFC, and high-temperature PEMFC, to name a few.
Modeling and simulation can be used to predict the current and potential distribution, the chemical species distribution, and the temperature distribution in a fuel cell. In this way, the cell can then be designed for the best possible utilization and operation for a given set of conditions. Important aspects are the removal of water and avoiding nonuniform utilization of the cell, which may result in poor performance and decreased lifespan. Additionally, microscopic aspects of the gas diffusion electrodes and the active layers can be studied, such as catalyst loading, particle size distribution, and bimodal pore distribution.